
This view was challenged in 1944, when Oswald Avery and colleagues demonstrated that bacterial DNA was likely the genetic agent that carried information from one organism to another in a process called "transformation". However, a compound of this kind, presumably common to all organisms, was considered too simple to hold the hereditary information known to reside in the chromosomes. The corresponding N-glycosides of the common sugar ribose are the building blocks of RNA, and are named adenosine, cytidine, guanosine and uridine (a thymidine analog missing the methyl group).įrom this evidence, nucleic acids may be formulated as alternating copolymers of phosphoric acid ( P) and nucleosides ( N), as shown: ~ P – N – P – N'– P – N''– P – N'''– P – N ~Īt first the four nucleosides, distinguished by prime marks in this crude formula, were assumed to be present in equal amounts, resulting in a uniform structure, such as that of starch. As noted in the 2'-deoxycytidine structure on the left, the numbering of the sugar carbons makes use of primed numbers to distinguish them from the heterocyclic base sites. The base components are colored green, and the sugar is black. Structures and names for these nucleosides will be displayed above by clicking on the heterocyclic base diagram. 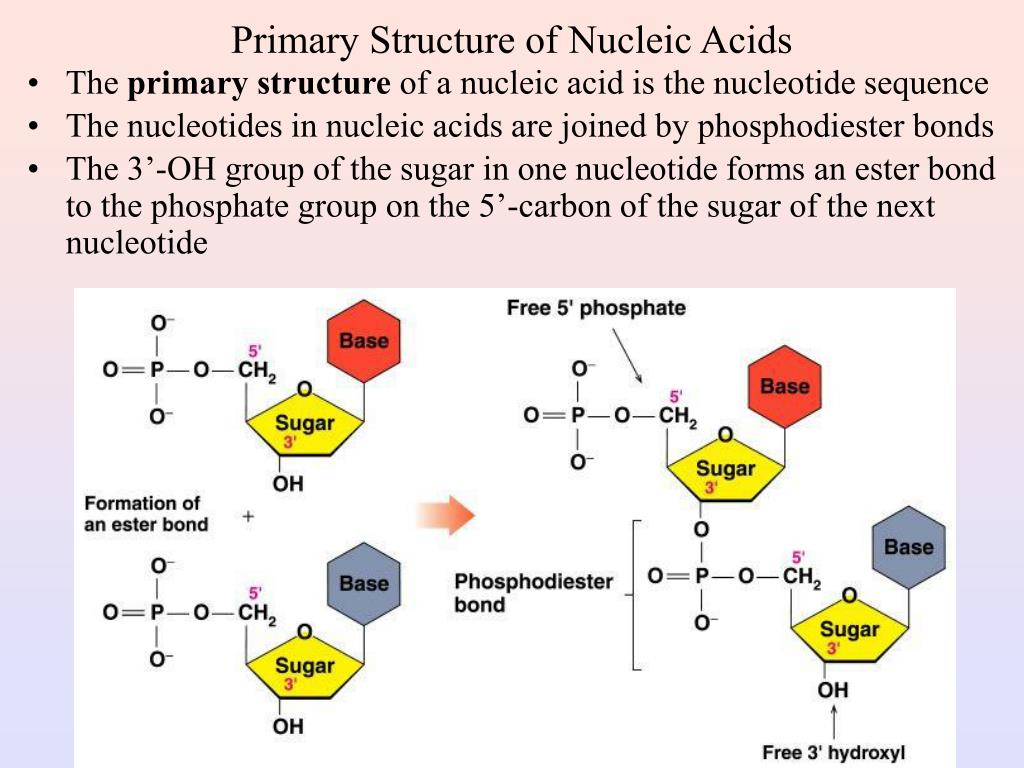
They are all polyfunctional bases, and may exist in tautomeric forms.īase-catalyzed hydrolysis of DNA gave four nucleoside products, which proved to be N-glycosides of 2'-deoxyribose combined with the heterocyclic amines. 
Each has at least one N-H site at which an organic substituent may be attached. The two monocyclic bases shown here are classified as pyrimidines, and the two bicyclic bases are purines. The acidic character of the nucleic acids was attributed to the phosphoric acid moiety. Analogous nucleic acids in which the sugar component is ribose are termed ribonucleic acids, abbreviated RNA. To reflect the unusual sugar component, chromosomal nucleic acids are called deoxyribonucleic acids, abbreviated DNA. Complete hydrolysis of chromosomal nucleic acids gave inorganic phosphate, 2-deoxyribose (a previously unknown sugar) and four different heterocyclic bases (shown in the following diagram). Unlike proteins, nucleic acids contained no sulfur. Elemental analysis of nucleic acids showed the presence of phosphorus, in addition to the usual C, H, N & O. In the 1920's nucleic acids were found to be major components of chromosomes, small gene-carrying bodies in the nuclei of complex cells. A few years later, Miescher separated nuclein into protein and nucleic acid components. He reported finding a weakly acidic substance of unknown function in the nuclei of human white blood cells, and named this material "nuclein". The first isolation of what we now refer to as DNA was accomplished by Johann Friedrich Miescher circa 1870. It should say the bonds holding the two DNA strands together are noncovalent hydrogen bonds.Nucleic Acids Nucleic Acids 1. It says "The nucleotides forming each DNA strand are connected by noncovalent bonds, called hydrogen bonds." but those are covalent peptide bonds. Ribose instead of deoxyribose is drawn in the dehydration synthesis diagram. These bonds are called 3’-5’ phosphodiester bonds"? So shouldn't it say "The phosphodiester bonds that join one DNA nucleotide to another always link the 3’ carbon of the first nucleotide to the 5’ carbon of the second nucleotide. These bonds are called 5’-3’ phosphodiester bonds." but DNA is synthesized from 5' to 3' direction according to the "How DNA is replicated" video where the 5'C of the second nucleotide is always linked to the 3'C of the first nucleotide. It says "The phosphodiester bonds that join one DNA nucleotide to another always link the 5’ carbon of the first nucleotide to the 3’ carbon of the second nucleotide. In the "nucleotide structure" diagram, deoxyribose is drawn but it says "ribose".
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
